Eco-friendly clinical disinfectant for ultimate performance
The OptiZil difference
| OptiZil | Other Surface Disinfectants | |
|---|---|---|
| 99.9999% efficacy (6 log) in under 1 minute | ||
| Powerful - eliminates biofilm & opportunistic pathogens | ||
| Non-irritating - pH neutral | Sometimes | |
| Wide use- spray or soak (cold sterilise) | ||
| Broadest spectrum of pathogen elimination | ||
| Zero Bioaccumulation risk | ||
| 100% Environmentally responsible | ||
| Versatile - spray or cold sterilisation |
Spray n’ Soak – for use on all dental surfaces. Can also be used for cold sterilising (soaking) alginates, impressions etc
OptiZil contains a naturally occurring antimicrobial (HOCl) that is versatile, safe, fast acting and proven to kill dangerous viruses, bacteria and spores. It is an extremely effective alcohol-free clinical disinfectant, acting in less than one minute on hard and soft surfaces. It can also be used safely on human skin and for cold sterilisation (soaking) of alginates, dentures and impressions.

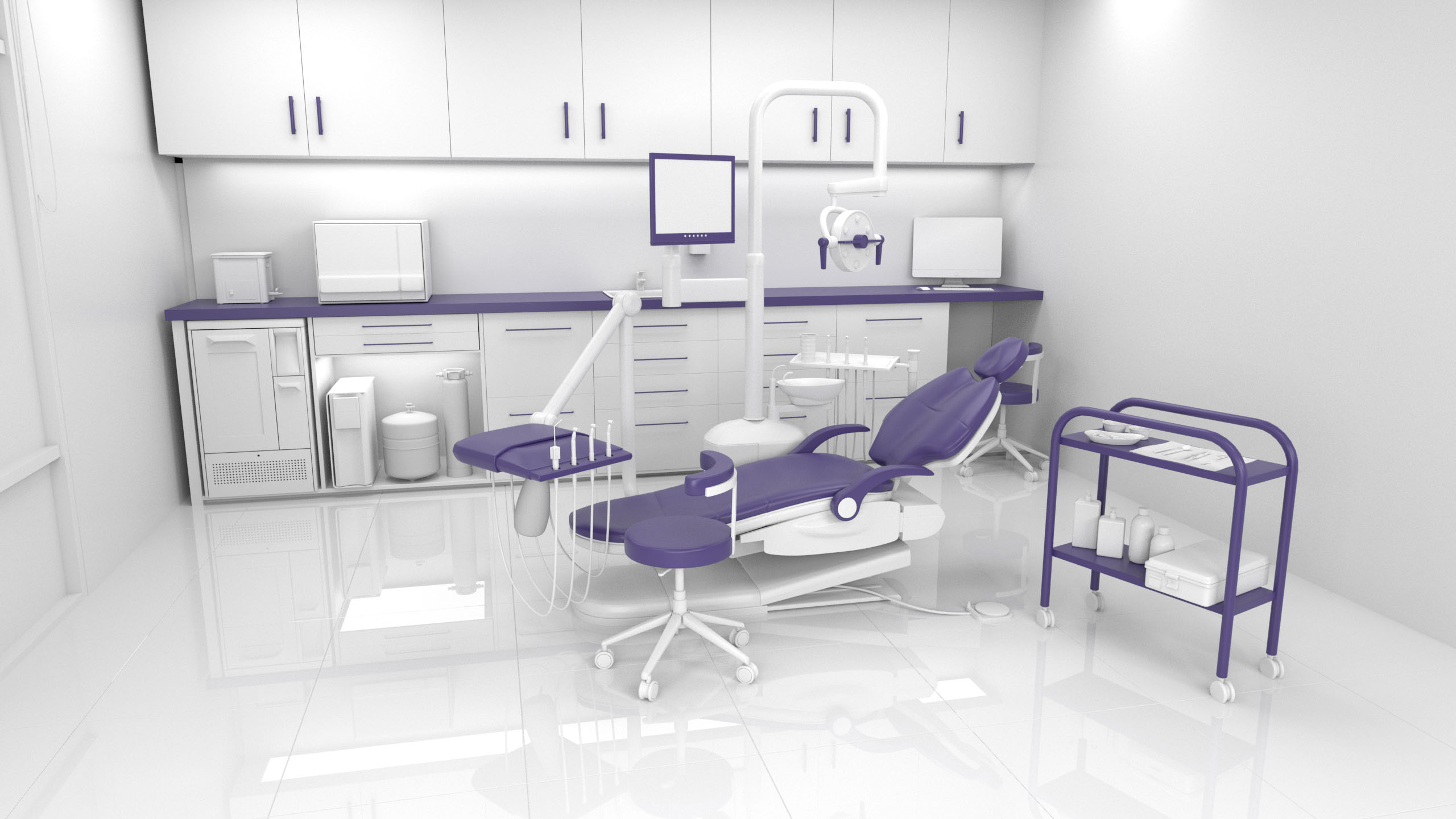
Know the Risks | Surface contamination can occur as a result of:

x
Reduce the risks | With OptiZil
Performance
-
Acts 5 times faster than traditional active ingredients, typically in less than 1 minute
-
80 times more effective than bleach
-
Effective on hard, soft and fabric surfaces and human skin
-
Stabilised for extended shelf-life
-
Deactivates prions & removes all five stages of biofilm growth
-
High quality dry wipes provide significant cost savings over traditional wet wipe alternatives
-
Convenient sizes: 100ml pump spray, 1L trigger spray and 5L containers
-
Saves money and reduces plastic waste
Safer
-
Ideal for high-volume use around the vulnerable and immunocompromised
-
Effective against enveloped viruses such as coronaviruses and Hepatitis B & C and non-enveloped viruses such as norovirus and parvovirus – BS EN 14476
-
Effective against healthcare-acquired infections such as Pseudomonas, E. Coli and MRSA
-
Free from quaternary ammonium compounds (QACs), bleach, alcohol and aldehydes
-
Biomimetic – uses the same chemical released in the body’s immune response
-
Hypoallergenic, pH neutral, non-cytotoxic and biodegradable
Log Reduction
OptiZil Knowledge
1. Testing & Regulatory Standards
- File Holder for the active ingredient (“Active chlorine generated from sodium chloride by electrolysis”) on ECHA’s Article 95 list of the Biocide Product Regulations (BPR)
- ISO16954 – Evaluating treatment methods intended to prevent or remove biofilm in dental unit procedural water delivery systems.
- BS EN 1040 – Quantitative suspension test for the evaluation of the basic bactericidal activity of chemical disinfectants/antiseptics. For chemical disinfectants and antiseptics.
- BS EN 1276 – Quantitative suspension test for the evaluation of the bactericidal activity of chemical disinfectants and antiseptics used in food, industrial, domestic and institutional areas.
- BS EN 13697 – Chemical disinfectants and antiseptics. Quantitative non-porous surface test for the evaluation of bactericidal and/or fungicidal activity of chemical disinfectants used in food, industrial, domestic and institutional areas
- BS EN 13704 – Chemical disinfectants. Quantitative suspension test for the evaluation of the sporicidal activity of chemical disinfectants used in food, industrial, domestic and institutional areas
- BS EN 13727 – Chemical disinfectants and antiseptics. Quantitative suspension test for the evaluation of bactericidal activity in the medical area
- BS EN 14204 – Chemical disinfectants and antiseptics. Quantitative suspension test for the evaluation of the mycobactericidal activity of chemical disinfectants and antiseptics used in the veterinary area
- BS EN 14348 – Chemical disinfectants and antiseptics. Quantitative suspension test for the evaluation of the mycobactericidal activity of chemical disinfectants in the medical area including instrument disinfectants
- BS EN 14476 – Chemical disinfectants and antiseptics. Quantitative suspension test for the evaluation of virucidal activity in the medical area
- BS EN 14561 – Chemical disinfectants and antiseptics. Quantitative carrier test for the evaluation of bactericidal activity for instruments used in the medical area.
- BS EN 14562 – Chemical disinfectants and antiseptics. Quantitative carrier test for the evaluation of fungicidal or yeasticidal activity for instruments used in the medical area
2. Evidence
- HOCl is a non-toxic, non-corrosive disinfectant against the virus that causes COVID-19: Block, MS and Rowan, BG. Hypochlorous Acid: A Review. The Journal of Oral and Maxillofacial Surgery, 2020
- The strong bactericidal ability of HOCl serves as a reliable defence disinfectant: Yan P et al. New Clinical Applications of Electrolyzed Water: A Review. Micro-organisms, 2021.